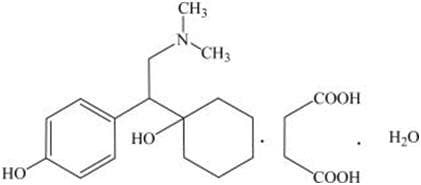
Desvenlafaxine Succinate
50 mg/1 · Tablet Film Coated Extended Release
By Lupin Pharmaceuticals, Inc.
- NDC
- 68180-592
- Route
- Oral
- Therapeutic class
- Cytochrome P450 2D6 Inhibitors [MoA], Norepinephrine Uptake Inhibitors [MoA], Serotonin Uptake Inhibitors [MoA], Serotonin and Norepinephrine Reuptake Inhibitor
- Marketing category
- ANDA
Package sizes
- 30 TABLET, FILM COATED, EXTENDED RELEASE in 1 BOTTLE (68180-592-06)NDC 68180-592-06Available to order
Additional information
Full prescribing information for this product is available on request. Submit a sourcing request and our team will share lot details, dating, and current pricing.
Request this medication
Send a request and we'll respond with availability, lot details, and pricing within 2 business hours.

