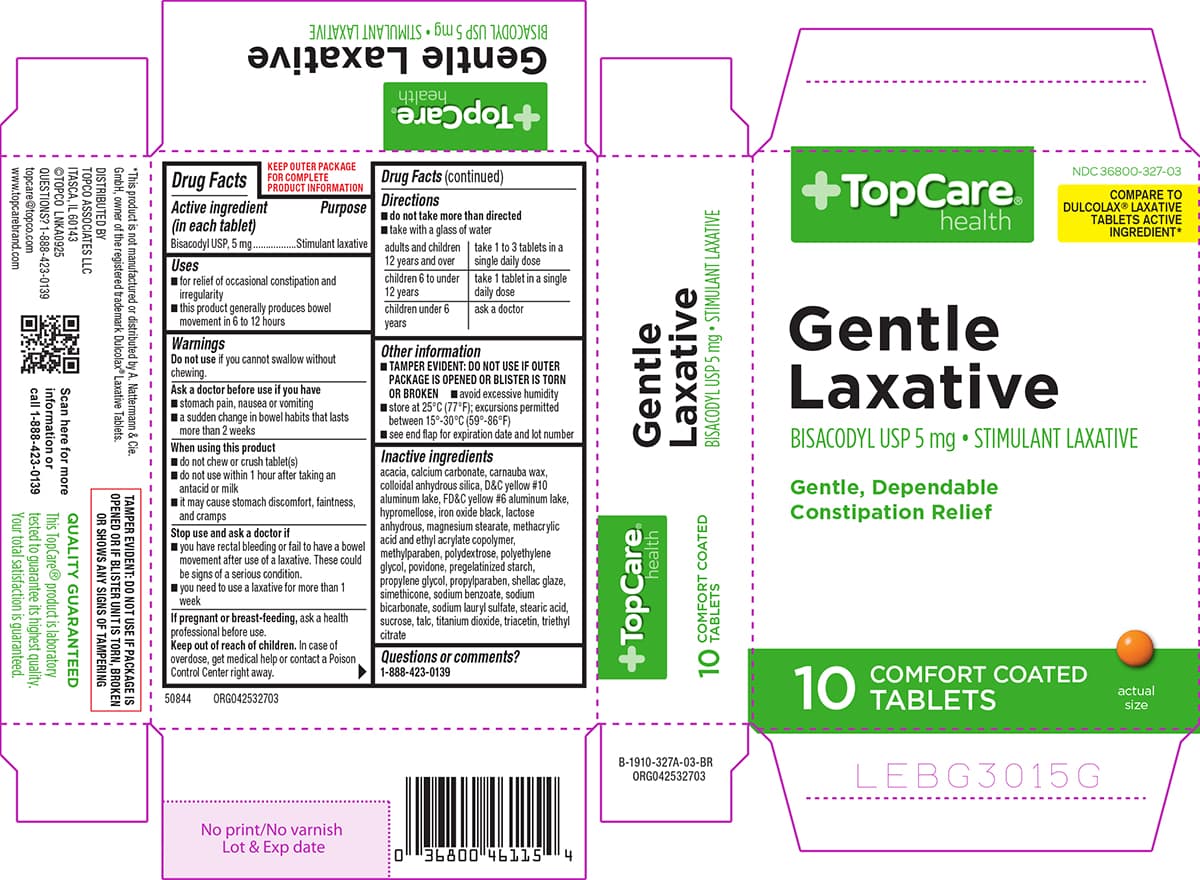
Bisacodyl
5 mg/1 · Delayed-Release Tablet
By Aphena Pharma Solutions - Tennessee, LLC
- NDC
- 71610-700
- Route
- Oral
- Therapeutic class
- Increased Large Intestinal Motility [PE], Stimulant Laxative , Stimulation Large Intestine Fluid/Electrolyte Secretion [PE]
- Marketing category
- OTC MONOGRAPH DRUG
Package sizes
- 2 TABLET, DELAYED RELEASE in 1 BOTTLE, PLASTIC (71610-700-02)NDC 71610-700-02Available to order
- 3 TABLET, DELAYED RELEASE in 1 BOTTLE, PLASTIC (71610-700-03)NDC 71610-700-03Available to order
- 4 TABLET, DELAYED RELEASE in 1 BOTTLE, PLASTIC (71610-700-04)NDC 71610-700-04Available to order
- 6 TABLET, DELAYED RELEASE in 1 BOTTLE, PLASTIC (71610-700-06)NDC 71610-700-06Available to order
- 8 TABLET, DELAYED RELEASE in 1 BOTTLE, PLASTIC (71610-700-08)NDC 71610-700-08Available to order
- 20 TABLET, DELAYED RELEASE in 1 BOTTLE, PLASTIC (71610-700-20)NDC 71610-700-20Available to order
Additional information
Full prescribing information for this product is available on request. Submit a sourcing request and our team will share lot details, dating, and current pricing.
Request this medication
Send a request and we'll respond with availability, lot details, and pricing within 2 business hours.
Related in Gastrointestinal
See all →Coppertone Sport Sunscreen SPF 30
2 g/100g / 8 g/100g +2 · Spray
Beiersdorf Inc
Available to source
Coppertone Tanning Sunscreen SPF 15 ((To Deliver) Avobenzone 2%, Octisalate 4.5%, Octocrylene 7%)
2 g/100g / 4.5 g/100g +1 · Aerosol
Beiersdorf Inc
Available to source
Coppertone Kids Sunscreen SPF 50
3 g/100g / 10 g/100g +2 · Aerosol Spray
Beiersdorf Inc
Available to source
Coppertone Sport Sunscreen SPF 50
3 g/100g / 10 g/100g +2 · Spray
Beiersdorf Inc
Available to source
